Before there was Sherlock Holmes and his trademark deerstalker cap, there was an English physician, Dr. Arthur Conan Doyle. And before that there was just plain old Arthur Conan Doyle, a student at the University of Edinburgh Medical School, and where we will begin.
On September 20th, 1879 the British Medical Journal published a letter entitled “Gelseminum as a Poison” in which Doyle recounts his use of a tincture of gelseminum (which we now call gelsemium) for successfully treating a case of neuralgia, or neuropathic (nerve) pain. Obviously prone to experimentation, the 19-year-old Doyle was “determined to ascertain how far one might go in taking the drug, and what the primary symptoms of an overdose might be.” Allrighty then. Doyle prepared a fresh tincture and recorded his observations like any good scientist should: he noted the dose and physiological effects, avoided tobacco, and dosed himself at the same time each day.
At low doses of 40 and 60 minums (1 minum = 1 drop = 0.06 mL) he observed no effects, but 20-minutes after ingesting 90 minums he experienced extreme “giddiness.” At 120 minums the giddiness was lessened, but several hours later he had vision problems. Psychological symptoms were gone by 150 minums, and all that remained were headaches and diarrhea. Most would have stopped here, but not our young man. Doyle pushed on towards 200 minum, at which the persistent and prostrating diarrhea, headache, and weak pulse got the better of him.
He concluded that healthy adults may take up to 90 minums, but that at doses of 90-120 the drug induces a sort of mild paralysis. Even higher doses bring upon unwanted side-effects, but he believed a person may become tolerant to its effects, much like opium. He signed off the letter with the bravado and gravitas we would expect from a man with a bit of Sherlock Holmes in him:
I feel convinced that I could have taken as much as half an ounce of the tincture, had it not been for the extreme diarrhoea it brought on.
-Believe me, yours sincerely,
A. C. D.
Clifton House, Aston Road, Birmingham.
The gelsemium Doyle referred to is a plant of the genus Gelsemium, which is home to three species: elegans, rankinii, and sempervirens. All are viney, climbing shrubs with small, trumpet-shaped, bright yellow flowers. Of the three, only Gelsemium elegans is found outside of the southeastern United States, and is native to China and southeast Asia. It’s probably a safe bet that the gelsemium obtained by the young Arthur Conan Doyle was Gelsemium rankinii, known as swamp jasmine, or Gelsemium sempervirens, known as Carolina Jasmine. Because I lived in Charleston, South Carolina for a few years, and witnessed Carolina Jasmine covering archways and walls, I’m going to dictate from here on out that Doyle obtained the sempervirens species. Not that it really matters, because chemically the two are nearly identical.
Like most natural products, Gelsemium is not comprised of just one chemical or alkaloid, it contains well over a hundred. But of those hundred plus chemicals, gelsemine has been found to be the most active, and received the most attention. Gelsemine’s mechanism of action is a bit complicated, but fascinating.
On the surface, gelsemine exhibits activity by way of the glycine receptors (2). If that sounds vaguely familiar, then you obviously read and retained a little bit from the strychnine post. The amino acid glycine is an inhibitory transmitter that controls, and slows down, the activity of motor neurons – nerve cells that start in the brain stem, project down the spinal cord, and controls muscle movement. In simplistic terms you can think of the glycine receptors like a governor on your car engine, which keeps it from going above a certain speed or RPM. Strychnine is an antagonist of the glycine receptor – it binds to the receptor more efficiently than glycine, yet doesn’t regulate it directly. Thus when exposed to strychnine, the motor neurons are firing wildly and out of control, resulting in seizures, and often death. Not good.
Gelsemine however, is an agonist. It mimics the actions of glycine, and slows the firing of motor neurons – the opposite of having a seizure. This activity probably accounts for young Doyle’s observation of a sort of mild paralysis. His motor neurons were slowed and he was experiencing a delay or inhibition of voluntary muscle movement. The diarhhea is likely the result of diminished involutary muscle control, probably of the internal anal sphincter, which keeps things where they should . . . ’til it’s time to go of course.
Muscle control was only part of Doyle’s descriptions, and simple glycine receptor pharmacology doesn’t fully explain the alleviation of his neuropathic pain, nor his “giddiness”, which I’ll translate to a general state of well being. This is where things get interesting and the fun begins. The game is afoot!
Discussing pain could take months, it’s that complex. I’ve got 50 words. There are mainly two types of pain: nociceptive and neuropathic. Nociceptive pain can be either chemical, mechanical, or thermal. Neuropathic pain is nerve related, with a pins-and-needles or burning sensation, brought about by allodynia – a fancy word meaning something that typically shouldn’t bring about pain, like a light touch. How’d I do? Forty-eight words? Awesome.
Gelsemine exhibits biological activity in animal models of neuropathic pain, whose actions are antagonized (reversed) by strychnine, so we know that the glycine receptors, and specifically the α3-glycine receptor is involved. Animal models have shown that activation of the α3-glycine receptor in the spinal cord stimulates the production of allopregnanolone, a neurosteroid that in a round-about-way is derived from cholesterol (3). Allopregnanolone has been shown to induce analgesia by way of T-type calcium channels. We’ve talked before about sodium channels with aconitine and saxitoxin, and the calcium channels work in similar ways and helps control muscle contraction, particularly those of the heart. Another role though, has been found, and that is with certain isoforms causing neuropathic pain, such as in diabetic neuropathy. (4, review article)
Another biomechanism may also be in play in decreased sensitivity to pain, and also explain the “well being” observed by Doyle, and that is activity on the GABA receptors. Allopregnanolone is a positive allosteric modulator of the GABA system – meaning that it amplifies the effects of GABA agonists, or “kicks it up a notch,” for all you Emeril Lagasse fans. GABAergic activity of allopregnanolone reduces anxiety and depression and may even be implicated in helping reduce the onset and symptoms of Alzheimer’s disease (5, review article). Anxiolytic drugs (anti-anxiety) that work on the GABA receptors, like the benzodiazepines Xanax and Valium (alprazolam and diazepam) are often (over)prescribed to people with chronic pain. Whether the pain is truly lessened, or just that the individual cares or stresses less about the pain, is debatable, but I vote for the latter.
The role of gelsemine, and gelsemium extracts, for use as an analgesic for chronic neuropathic pain and as an anxiolytic is supported by recent pharmacological studies, and is the subject of a lot of current research efforts. As a pharmaceutical target, I’d rank it as “interesting” and probably worthy of some time researching, but the 25-step, 1% overall yield synthesis scares me (6).
Our young Arthur Conan Doyle was not the first, nor the last, to explore gelsemium extracts, but it gave future scientists 100-years later a clue. Pharmaceutical research is not unlike being a detective, you are constantly searching for clues and formulating explanations for whatever results might be generated. Sherlock Holmes, and his drug-dabbling ways, did not make an appearance for another 8 years, but they are evident in Doyle’s exploits in self-poisoning and becoming a pharmaceutical detective.
References:
1. Doyle, A.C. “Gelseminum as a Poison.” British Medical Journal 2.977 (1879): 483.
2. Zhang, Jing-Yang, Nian Gong, Jin-Lu Huang, Ling-Chen Guo, and Yong-Xiang Wang. “Gelsemine, a Principal Alkaloid from Gelsemium Sempervirens Ait., Exhibits Potent and Specific Antinociception in Chronic Pain by Acting at Spinal α3 Glycine Receptors.” Pain® 154.11 (2013): 2452-462.
3. Venard, C., N. Boujedaini, P. Belon, A.g. Mensah-Nyagan, and C. Patte-Mensah. “Regulation of Neurosteroid Allopregnanolone Biosynthesis in the Rat Spinal Cord by Glycine and the Alkaloidal Analogs Strychnine and Gelsemine.” Neuroscience 153.1 (2008): 154-61.
4. Patte-Mensah, C., L. Meyer, O. Taleb, and A.g. Mensah-Nyagan. “Potential Role of Allopregnanolone for a Safe and Effective Therapy of Neuropathic Pain.”Progress in Neurobiology 113 (2014): 70-78.
5. Schüle, Cornelius, Caroline Nothdurfter, and Rainer Rupprecht. “The Role of Allopregnanolone in Depression and Anxiety.” Progress in Neurobiology113 (2014): 79-87.
6. Zhou, Xuan, Tao Xiao, Yusuke Iwama, and Yong Qin. “Biomimetic Total Synthesis of (+)-Gelsemine.” Angewandte Chemie International Edition51.20 (2012): 4909-912.


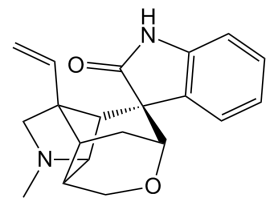
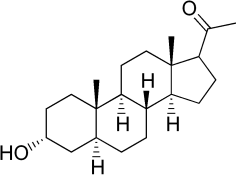
Diarrhoea instead of diarrhea, right?
Depends on what side of the Pond you’re on I guess. Brits like to add extra vowels and Yanks like to take them away.
But I will say, without spell-check I’m screwed. Diarrhea and nausea are the two words I never spell correctly.
Thanks for reading!
Going by the side Conan Doyle was on, I’d say he spelled it with an o.
Or Daria for Beavis and Butthead fans like me. Hahahaha!
Pingback: Tetrodotoxin: The Paralytic Pufferfish Poison | Nature's Poisons
Pingback: Episode 63: Con Drop – Three Patch Podcast
Pingback: 63 – Live from 221B Con: Drugs and Poisons – The Three Patch Podcast Casefiles
Pingback: El extraño caso de las flores amarillas – espacio de invernada
Pingback: For a long life, avoid the garden – Plant Cuttings